Laser Technology
Published: 12/11/2015
ACFS Offers Laser Assisted Hatching (LAH)
The advent of the laser has enabled the development of precision techniques to manipulate embryos for enhanced fertility. Laser-assisted hatching (LAH) can potentially enhance implantation/pregnancy rates, or reverse a history of failed embryo implantation into the uterine wall.
The specialists at Arizona Center for Fertility Studies strongly believe that laser technology is a state-of-the-art technology for any embryos that undergo Assisted Hatching (AH), embryo biopsy for Chromosome Abnormality / Single Gene Mutations or Gender or Sex Selection, and for pre-transfer of Cryopreserved or Frozen Embryos (FET).
Laser-assisted hatching (LAH) is used to help the embryo hatch from its protective outer shell (the zona pellucida) and promote implantation in the uterine wall after embryo transfer. Laser-assisted hatching uses a highly focused infrared laser beam to remove a tiny section of the zona pellucida, or outer wall of an embryo, in very precise increments. Prior to the clinical availability of laser-assisted hatching, only mechanical or chemical methods could be used for assisted hatching of human embryos in clinical settings. Laser-assisted hatching requires less handling of the embryo than these other assisted hatching methods. Also, laser-assisted hatching is faster than the other methods and, therefore, the embryo spends less time outside the incubator.
Picture of a day 3 eight-cell embryo about to undergo laser-assisted hatching (arrow, round marker, indicates where the laser beam will penetrate the zona pellucida)
Indications for laser-assisted hatching (LAH)
Laser-assisted hatching (LAH) is not currently recommended for routine use in all in-vitro fertilization patients. Factors that should be considered when determining laser-assisted hatching candidacy include: age, hormonal status, embryo quality, number of in-vitro fertilization attempts, and whether frozen embryos are being implanted.
- Age - Women over 37 years old - Arizona Center for Fertility Studies specialists believe that all embryos, whether day 3 or blastocysts, should undergo laser-assisted hatching to improve implantation/pregnancy rates
- Hormonal Status - Women with an elevated baseline level of FSH
- Embryo Quality - Women with poor prognosis embryos, including conditions such as a thick zona pellucida, slow cell division rate, or high-cell fragmentation
- In-Vitro Fertilization Attempts - Women who have failed 1 or more In-Vitro Fertilization cycles. We believe that laser-assisted hatching should be done on the first In-Vitro Fertilization attempt so a second attempt may not be needed
- Frozen Embryos - Women using either day 3 or blastocyst frozen/thawed embryos, which may have a hardened zona pellucida as a result of the freezing process. Arizona Center for Fertility Studies does laser-assisted hatching on all frozen embryo transfers (FET)
Laser-Assisted Biopsy (LAB)
Laser-Assisted Biopsy (LAB) is used at Arizona Center for Fertility Studies to facilitate the removal of a cell or blastomere from an embryo undergoing pre-implantation genetic diagnosis (PGD/PGS) for chromosome testing, single gene mutation testing or gender/sex selection. We use two methods for embryo biopsy:
- Blastomere Biopsy
- Trophectoderm Biopsy.
Blastomere Biopsy
Blastomere biopsy, the most common embryo biopsy method, involves the removal of one blastomere when the embryo reaches the eight-cell stage, typically on the third day of development.

Video of Arizona Center for Fertility Studies embryologist doing assisted hatching using the laser to remove a single cell or blastomere for chromosome testing and sex selection.
Trophectoderm Biopsy
Trophectoderm biopsy is gaining popularity as an alternative method of embryo biopsy. Since trophectoderm cells are extra-embryonic tissue, they become not part of the fetus, but part of supporting structures, such as the placenta and membranes. Trophectoderm biopsy takes place at the blastocyst (day 5 or 6) stage of development, as the trophectoderm is beginning to herniate through the zona pellucida. Instead of removing an individual blastomere or cell, we remove several trophectoderm cells.

Video of Arizona Center for Fertility Studies embryologist doing a trophectoderm biopsy using the laser for chromosome testing and sex selection
Our experience doing 23-chromosome microarray over the last several years has yielded what seems to be conclusive evidence that embryos can "self-correct". We strongly recommend only doing biopsies on day 5-6 embryos (blastocysts) using laser-directed TE (trophectoderm) biopsy. This means that after the blastocyst is biopsied, it will undergo cryopreservation and be transferred in a subsequent frozen embryo transfer (FET) the following cycle. Although this means that the couple will need to wait for a cycle to do the transfer, there are a number of advantages of doing a TE biopsy as opposed to a day 3 biopsy.
- First and foremost, it eliminates dealing with the possibility of "self-correction", which we’ve observed happens more commonly than would be expected. Arizona Center for Fertility Studies was in a unique situation to be able to re-biopsy normal-appearing day 5-6 blastocysts at no charge. Most clinics are not able to do this. If they are doing a day 3 biopsy and the chromosomes come back abnormal, they have to discard the embryos because without re-biopsy there is no way to know if they "self-corrected" and it’s too risky to just assume they did. If a patient feels strongly that she wants to do a day 3 biopsy, in order to avoid a FET and has developmentally normal blastocysts that came back chromosomally abnormal, Arizona Center for Fertility Studies will still do a TE biopsy, at no charge, and cryopreserve the embryo(s), then wait for TE biopsy results. If the re-biopsy is normal, she will be able to do a future FET if needed. Although we strongly recommend day 5-6 TE biopsy for PGT with subsequent FET, couples do not have to worry about discarding "normal" embryos.
- A day 5-6 laser-directed TE biopsy is optimal because evidence suggests that embryos do better in group culture through the blastocyst stage than in isolation on day 3 (which is required for day 3 biopsies).
- Day 3 biopsy offers an increased chance of failed amplification (see below) because only one cell is being removed. With a TE biopsy, you can safely remove 3-4 cells, eliminating the risk of failed amplification and allowing more accurate interpretation.
- A TE biopsy and cryopreservation with subsequent FET can stimulate a patient more aggressively to make more eggs/embryos and be triggered with Lupron, rather than with hCG, completely avoiding the risk of ovarian hyperstimulation syndrome (OHSS).
- A couple only pays for TE biopsy if the embryos reach the blastocyst stage. If a day 3 biopsy is done, all embryos need to undergo a biopsy, since you do not know which ones will develop into blastocysts. With TE biopsy, if the embryos do not reach the blastocyst stage, which is of course not a good sign, at least you are not paying to have PGT done.
In the Arizona Center for Fertility Studies team’s experience, all these advantages collectively produce higher success rates when using PGT, which is why we strongly recommend that all patients undergoing PGT only do TE biopsy, cryopreservation, and subsequent next-cycle FET. Our out-of-state patients are the one exception unless they can make a return trip. Although our success rates are still very good with day 3 biopsy, we prefer day 5 TE biopsy.
Indications for Laser-Assisted Embryo Biopsy (LAB):
Laser-Assisted Embryo Biopsy (LAB) is not currently recommended for routine use in all in-vitro fertilization patients at the Arizona Center for Fertility Studies. Several factors should be considered in determining LAB candidacy. We strongly believe that it should be done on all embryos for the following indications:
- Carriers of Single Gene Disorders (genetic disorders like cystic fibrosis, muscular dystrophy, sickle cell anemia, Rh factor and many more) — these patients benefit from PGD/PGS by reducing the risk of conceiving an affected baby
- Recurrent Miscarriage - Fertile couples with repeated miscarriages (RPL) should be evaluated for the presence of a chromosomal abnormality — the female or male partner may be a carrier of a balanced translocation or be an aneuploid mosaic
- Unsuccessful In-Vitro Fertilization (IVF) Cycles - couples with repeated unsuccessful In-Vitro Fertilization (IVF) cycles should be evaluated for the presence of a chromosome abnormality — the female or male partner may be a carrier of a balanced translocation or be an aneuploid mosaic
- Unexplained Infertility - the most probable cause of unexplained infertility or history of repeated pregnancy loss is a chromosome abnormality in which the male or female partner may be a carrier of translocation or be an aneuploid mosaic
- Aneuploidy (embryos with chromosome abnormalities that result in a miscarriage or unsuccessful attempt at In-Vitro Fertilization (most commonly Down's Syndrome) or Advanced maternal Age - women of advanced maternal age (> 35) are at a higher risk of producing aneuploid embryos, resulting in implantation failure, a higher risk of miscarriage or the birth of a child with a chromosome abnormality
- Gender or sex selection - Couples wanting to determine the sex of each of their embryos for gender selection
Is Laser Technology Safe?
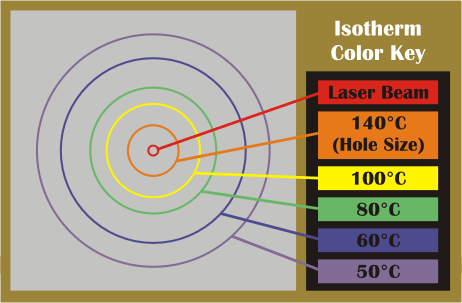
Yes - because the patented Isotherm Rings™ on our laser prevent the potentially harmful effects of laser drilling heat on blastomeres (the cells of the embryo) adjacent to the zona pellucida (wall of the embryo).
These Isotherm Rings™ appear on our embryologist's computer screen as a series of six concentric circles of varying colors (below) and indicate the maximum temperature reached the ring’s diameter at various laser pulse durations (2nd picture below). At longer pulse duration, it is apparent that temperatures radiate farther into the center of the embryo, increasing the likelihood of blastomere damage. Our Arizona Center for Fertility Studies embryologist uses the orange ring (second from the center) as an indicator of the drill hole size at the selected pulse duration.
With this interactive Isotherm Rings™ feature, our team can "see" the heating and drill hole size, eliminating the temperature "guesswork."

The heat conduction into the embryo, as shown by the Isotherm Rings™, is based on published scientific algorithms that are built into the software. For increased embryo safety, the Isotherm Rings™ are hardwired and their specifications cannot be altered or deleted by users. With the Isotherm Rings™ you can be confident that what you see is accurate and determined according to scientific definitions, not by individual preference.
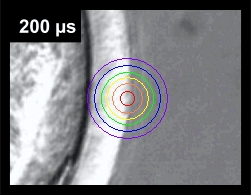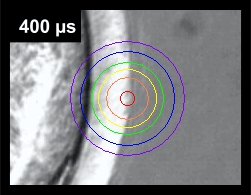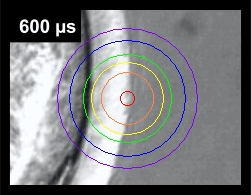
Compare the degree of heat penetration versus the drill hole size (orange ring) in the following animation that shows the degree of heat radiation at varying pulse durations.
Note how the heat penetrates further into the embryo as the pulse length increases. So even if the actual drill hole (orange ring) does not come close to the blastomeres, the blastomeres are exposed to the heat generated by the laser beam at longer pulse lengths. By using the Isotherm Rings™ feature of our laser, our embryologist can "see" the heat conduction prior to treatment and adjust the pulse length and embryo positioning accordingly, so as NOT to damage the embryo.
We’re excited about offering this new technology in human reproduction that has become the standard at our clinic.
Pictures and some text - Courtesy of Hamilton Thorne











